Services informatiques et intégration
Un service compétent et des interfaces de communication fiables
Les appareils de diagnostic Otopront peuvent être facilement intégrés dans des réseaux - qu'il s'agisse de l'ordinateur de votre cabinet ou de réseaux HIS complexes. Un logiciel de base commun (Otopront App Manager), notre déclaration de conformité DICOM et notre équipe informatique expérimentée permettent un processus rapide et simple.
L'intégration dans l'ordinateur du cabinet est généralement réalisée en ligne par télémaintenance par notre service informatique après la livraison de l'appareil. Entre-temps, les questions en suspens concernant le logiciel du cabinet et les exigences particulières sont clarifiées.
Grâce à notre conformité DICOM, les appareils d'imagerie (par exemple PES PILOT HDpro, PES PILOT HDpro Stroboskop) peuvent être standardisés dans les réseaux HIS. La société INNOFORCE nous soutient dans la mise en œuvre.
L'intégration au réseau en un coup d'œil
Installation facile dans l'ordinateur du cabinet (par exemple Medistar, Turbomed, Albis, tomedo etc.) via le protocole GDT
Reconnaissance automatique de la commande par l'ordinateur du cabinet, ou par simple pression d'un bouton
Intégration complète dans les SIH et PACS via des interfaces standard (HL7, compatible DICOM)
En option, transfert de données vidéo, communication avec la liste de travail (par exemple, DICOM vidéo, DICOM liste de travail).Déclaration de conformité DICOM
IT service
Notre service informatique veille en permanence à ce que votre intégration soit réalisée rapidement et que vos applications logicielles fonctionnent toujours correctement. Si quelque chose ne fonctionne pas, notre personnel informatique se concentrera sur un dépannage rapide et pragmatique. Notre objectif est de vous fournir une qualité de service de premier ordre, ainsi que l'entretien de votre appareil.
Le logiciel de télémaintenance sur nos appareils permet un service en ligne rapide, sans devoir convenir d'une date de visite coûteuse. En outre, les mises à jour du logiciel peuvent également être installées de cette manière.
Intégration dans la pratique EDP
Le programme de base (Otopront App Manager) installé sur tous les appareils Otopront dispose d'une interface GDT standardisée, ce qui permet d'intégrer facilement les appareils dans l'EDP de votre cabinet et de communiquer les résultats.
A titre d'exemple, l'intégration unique est la suivante :
1. Informations sur l'EDP de la pratique fournies à Otopront (par exemple, type : Medistar, Turbomed, Albis, etc.)
2. Intégration par télémaintenance à la date convenue ; environ 2 à 3 heures de travail.

Intégration dans les HIS (systèmes d'information hospitaliers)
L'intégration des appareils Otopront dans les systèmes HIS et PACS se fait en coopération avec INNOFORCE, un spécialiste des réseaux et de l'administration des données des patients dans les hôpitaux.
En plus d'une intégration standardisée via DICOM, HL7 est utilisé comme protocole supplémentaire. La portée fonctionnelle souhaitée (par exemple, archivage dans le PACS, listes de travail DICOM) peut être définie de manière variable et des demandes spéciales peuvent être prises en compte. Après avoir rempli un questionnaire standardisé et une préparation interne, l'intégration peut généralement avoir lieu en quelques heures sur place.
Nous serions heureux de partager avec vous des cliniques de référence ayant intégré Otopront, afin que vous puissiez vous faire une idée plus précise du processus et de l'organisation de notre intégration.

Intégration DICOM du PES PILOT HDpro
Gestion des patients et des résultats
Otopront App Manager
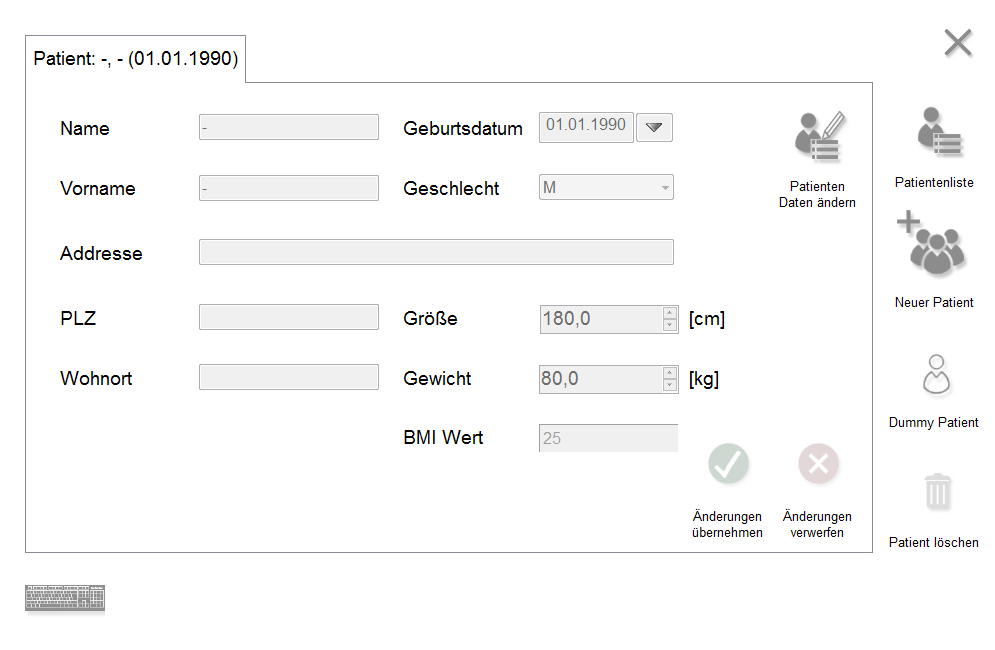
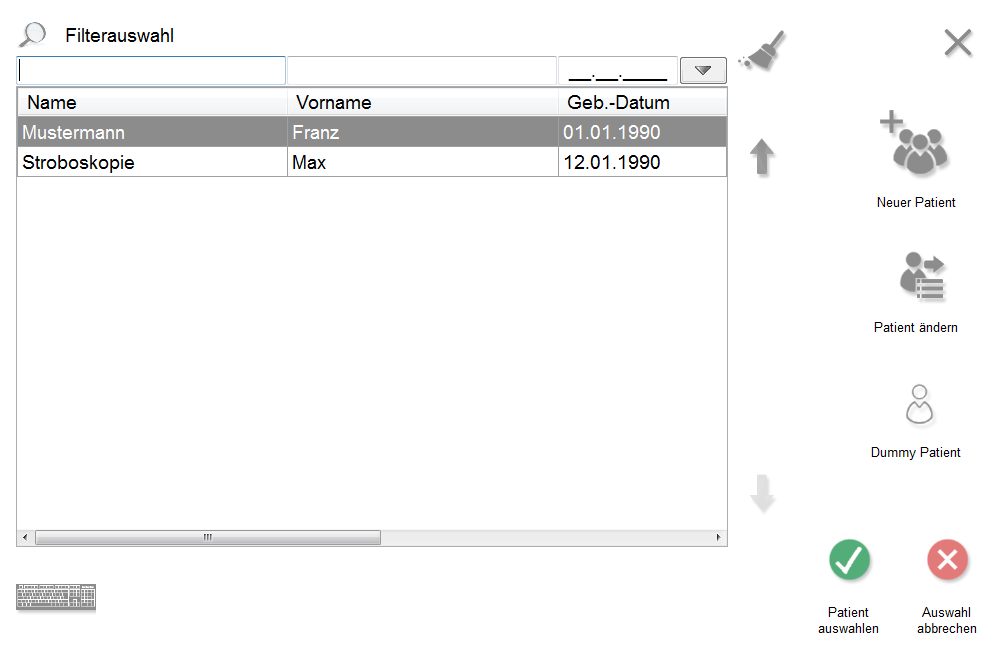
Notre gestionnaire d'applications est un logiciel de base sur lequel sont basées toutes les autres applications de modules pour nos appareils. Il est installé sur chaque système et dispose d'une gestion complète des patients et des résultats, qui peut être partagée par tous les appareils. Ainsi, les différents résultats diagnostiques d'un patient peuvent être obtenus, par exemple, à partir de DIGITAL 5, RHINO-SYS et PES PILOT HDpro sur un PC.
Avec l'App Manager comme base commune avec l'interface GDT, chaque appareil peut être intégré de manière identique - ce qui réduit le temps et les efforts et simplifie tout travail de maintenance.

INNOFORCE Responsable du diagnostic
L'assistant optionnel pour gérer vos enquêtes
Diagnostic Manager est un programme de base de données sophistiqué pour les cabinets d'ORL, dans lequel les résultats des différentes procédures de mesure sont visualisés et stockés. Vous pouvez rapidement et facilement récupérer les données souhaitées à partir de tous les postes de travail et les imprimer si nécessaire. Le Diagnostic Manager peut être intégré dans un programme de cabinet existant (par exemple, Turbomed, Medistar, etc.) via l'interface GDT. Cela vous permet de récupérer et d'afficher les résultats de mesure souhaités directement à partir du programme du cabinet.
Diagnostic Manager s'appuie sur le savoir-faire d'INNOFORCE avec le programme de base de données ENTstatistics, qui est utilisé avec succès depuis 2006. La technologie de serveur MSSQL utilisée est puissante et largement acceptée pour les applications en cabinet et à l'hôpital. Diagnostic Manager est un programme supplémentaire ouvert et peut gérer non seulement vos produits Otopront mais aussi les données des appareils de diagnostic d'autres fabricants.





